Features
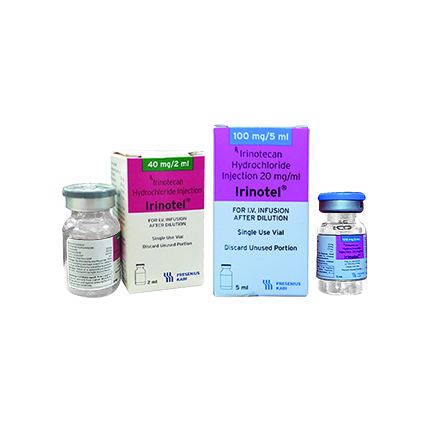
The agent Irinotecan is a cytostatic and belongs to the therapeutic class of Topoisomerase I inhibitors.
Irinotecan is a derivative of camptothecin. Camptothecins interact specifically with the enzyme topoisomerase I which relieves torsional strain in DNA by inducing reversible single-strand breaks. Irinotecan and its active metabolite SN-38 bind to the topoisomerase I-DNA complex and prevent religation of these single-strand breaks. Current research suggests that the cytotoxicity of irinotecan is due to double-strand DNA damage produced during DNA synthesis when replication enzymes interact with the ternary complex formed by topoisomerase I, DNA, and either irinotecan or SN-38. Mammalian cells cannot efficiently repair these double-strand breaks.